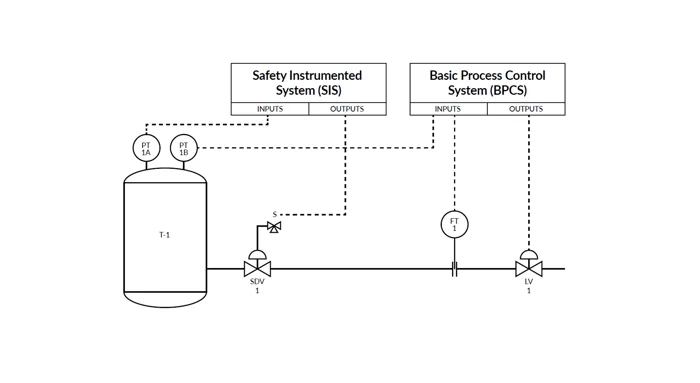
Patient safety, compliance and data integrity
In pharmaceutical manufacturing it’s essential that strict safety guidelines and regulations are followed, and manufacturing processes are consistent and reliable. This safeguards product purity and ensures the highest possible end-product quality, which is vital to maintaining patient safety.
Pharmaceutical manufacturers require a calibration ecosystem that complies with stringent industry regulations while delivering productivity gains and improving data integrity and quality standards.
Experience a better way to manufacture life-saving products
Many of the world’s leading pharmaceutical and life sciences companies rely on the Beamex calibration ecosystem, all designed to help customers achieve their calibration-related goals in line with regulations, such as those from the FDA and MHRA.
Our calibration solutions have been developed for more than 50 years by combining our own experience with feedback gained through close cooperation with household names in the pharmaceutical industry.

Pharmaceutical industry insights

Tackling challenges in the pharmaceutical industry through digitalization
Patient safety and ensuring compliance with regulations -including FDA Current Good Manufacturing Practice (CGMP) and 21 CFR part 11 – are the top priorities.
Read article >

Adapting to trends in the pharmaceutical industry
Recent trends in the research-based pharmaceuticals industry, including personalized medicine, flexible production facilities, and digitalization, are impacting established processes and require new ways of thinking.
Read article >

How a trusted advisor can help maximize return on digital investments
Many players in the pharma industry are making investments in digital solutions in order to enhance their calibration processes. But how can you make sure these investments pay off and do not negatively affect compliance or patient safety?
Read article >
Discover how much you could save on operational costs with the calibration process savings calculator.
Customer success stories
300 hr
Reduction in time spent on calibrations annually
GlaxoSmith Kline Ltd, Ireland
>40%
Reduction in time and resources needed to perform calibrations
Glatt Air Techniques, US
What our pharmaceutical customers say
Are you performing calibrations? Browse all our “how to” calibrate content and discover better ways to calibrate.
Beamex calibration solutions

Calibration management
Discover how Beamex calibration software helps you digitalize and automate calibration processes.
Learn more >

Field calibration
Experience the Beamex portfolio of advanced field calibration technology.
Learn more >

Workshop calibration
Meet the Beamex range of industrial workshop calibration solutions.
Learn more >

Services
Get the most out of Beamex technology with expert services, and calibration and repair services.
Learn more >
Together we can find the ideal calibration solution for your operations

Pharmaceutical User Forum
User meetings are an opportunity to gain powerful insights into working practices and exchange ideas for new products and services. In the Pharmaceutical User Forum industry leaders share experiences and challenges, contribute significantly to product development and learn about new products in advance.
Novartis, GlaxoSmithKline, Boehringer-Ingelheim, Astellas, AstraZeneca and Orion are just some of the pharmaceutical companies that have participated in these meetings.
Would you like to know more about the Pharmaceutical User Forum or are you interested in joining the next meeting, then contact us.